Quercus falcata Michx. - Southern Red Oak, Spanish Oak
Common Names
Southern Red Oak, Spanish OakField Identification
Large deciduous tree with alternate, simple, lobed leaves; with pendant catkin-like flowers in early summer followed by acorns in the fall.Medicinal uses
Used by Native Americans to treat a variety of maladies including dysentery, indigestion, mouth sores, chapped skin, fevers, asthma, and urinary tract disorders. (Moerman, 1998)Other uses
The Native Americans used the fiber for constructing baskets, wood for firewood as well as making tools, and leaves for wrapping dough in bread making. The wood was used by European settlers for making lumber, railroad ties, wagon wheels, and furniture. (Moerman, 1998)Oak-mast surveys may help wildlife agencies better understand dynamics of fall harvests and may be useful in harvest management models that attempt to stabilize fall harvest rates of game animals (Norman, 2003).
Tannins derived from oaks have been used historically to tan animal hides into leather. (Burrows & Tyrl, 2001)
Poisonous Properties
Oak leaves, buds, bark, and acorns contain tannins which have varying degrees of toxicity in different animals. Although oak foliage and acorns provide valuable food for many wildlife species and even some livestock, oak toxicosis, a urinary and digestive tract disease can occur when some animals are forced to subsist on oaks exclusively for several days. Poisoning is rare in humans due to the large amounts needed to ingest to cause symptoms. (Burrows & Tyrl, 2001)
Stories
Dryads (or "tree spirits") are nymphs associated with Greek mythology which live near, or in, trees. Dryads are born bonded to a specific tree, originally, in the Indo-European Celtic-Druidic culture, an oak tree. Drys in Greek signifies 'oak,' from an Indo-European root *derew(o)- 'tree' or 'wood.'
In a nationwide vote hosted by the National Arbor Day Foundation in 2001, the oak was selected as America's National Tree.
Nomenclature
Quercus falcata Michx., Hist. Chenes Amer., no. 16, pl. 28. 1801.
*Quercus rubra L., Sp. Pl. 996. 1753. p.p.
Quercus nigra digitata Marsh., Arbust. Am. 121. 1785.
?Quercus rubra montana Marsh., Arbust. Am. 123. 1785.
Quercus elongata Muhl. in Willd., Neu. Schrift. Naturf. Fr. Berlin 3: 400. 1801.
Quercus triloba Michx., Hist. Chenes Amer., no. 14, pl. 26. 1801.
Quercus falcata var. triloba Nutt., Gen. N. Am. Pl. 2: 214. 1818.
Quercus aurea Raf., Alsogr. Am. 20. 1838.
Quercus discolor Spach, Hist. Nat. Veg. Phan. 11: 163. 1842. not Ait. 1789.
Quercus falcata var. ludoviciana A. de Cand., Prodr. 16,2: 59. 1864.
Quercus hypophaeos Kirch. in Petzold & Kirch., Arb. Muscav. 656. 1864.
Quercus nobilis hort. ex K. Koch, Dendrol. 2,2: 65. 1872.
Quercus digitata (Marsh.) Sudw., Gard. & For. 5: 99. 1892.
Quercus cuneata Dipp., Handb. Laubh. 2: 111, f. 52. 1892.
Quercus cuneata var. falcata Dipp., Laubh. 2: 111, f. 53. 1892.
Quercus rubra triloba (Michx.) Ashe, Proc. Soc. Am. For. 11: 90. 1916.
Quercus rubra f. cuneata (Dipp.) Trel., Mem. Nat. Acad. Sci. 20: 202. 1924.
Description
HABIT Perennial, deciduous, phanerophytic, tree, diclinous and monoecious, 25-30 m tall.
STEMS Main stems ascending or erect, round. Bark dark brown or dark gray, tightly irregularly fissured with scaly ridges, not exfoliating. One study found bark roughness increasing from mesic to xeric sites (Glitzenstein & Harcombe, 1979). Branches erect or ascending or horizontal. Twigs dark brown or brown or gray, fluted-terete, 2-5 mm in diameter, smooth and lenticellate. 1st year twigs densely covered with fasciculate erect or spreading brown to light brown hairs, hairs becoming less dense and turning light gray on 2nd-3rd year twigs; glabrous on older twigs. Pith white, 5-pointed, continuous, nodal diaphragm absent. Sap translucent. For an anatomical study of the xylem see Tillson & Muller, 1942.
BUDS Terminal and axillary present, clustered at twig apices and scattered along stem, usually densely brown or gray hairy for most of their length. Terminal bud ovoid, semi-pointed; axillary buds ovoid, semi-pointed. Bud scales dark reddish-brown or brown, imbricate, with short and unbranched appressed brown or light brown hairs, sparsely to moderately distributed. Bud scale scars encircling the twig. Leaf scars crescent or half-round shaped. Vascular bundle scars numerous, scattered.
LEAVES Alternate, simple, (appearing pseudo-opposite or pseudo-whorled at twig apices), crowded toward stem apex or spaced somewhat evenly along and divergent from stem. Stipules lateral, free from the petiole, linear, caducous. Petiole flattened adaxially, 1.5-6 cm long, usually with brown to light brown fasciculate erect to spreading hairs, moderately densely or densely distributed throughout (rarely glabrous on some late season petioles). Leaf blades: abaxial surface light yellowish green to yellowish green or light green to light gray-green; adaxial surface green to dark green and usually glossy; elliptic or ovate or obovate, asymmetric or bilaterally symmetric, 8-25 cm long, 5-13 cm wide, chartaceous; base obtuse or U-shaped, occasionally semi-cuneate and occasionally oblique; margins sometimes symmetrically, more often asymmetrically 1-4 lobed per side (var. triloba usually only 3-lobed distally), 1/4-3/4 distance to the midvein with rounded sinuses and acute aristate apices; terminal lobe often elongated and falcate with acute aristate apex. Abaxial surface usually exhibits the following hair types: a. fasciculate brown to light brown erect to spreading hairs, moderately distributed along the main veins; b. simple brown to light brown erect hairs, sparsely to moderately distributed along the main veins; c. minute simple yellowish more or less appressed multi-cellular glandular hairs, sparsely to moderately distributed throughout; d. tufted multi-radiate light gray (occasionally light brown) hairs densely distributed throughout. Adaxial surface with all aforementioned hair types when young, retaining only sparsely scattered simple and multi-radiate hairs when mature (these minute, surface appearing glabrous with naked eye), see Hardin, 1979a;Thompson & Mohlenbrock, 1979; and for an overview of the phenology and the variability and density of hairs due to ecological factors and hybridization see Hardin, 1979b. For a study of morphological variation see Jensen, 1989.
FEMALE INFLORESCENCES Coetaneous, spike consisting of a single flower (sometimes 2-3), in axils of current year leaves, subsessile initially, often becoming short-pedunculate in fruit, surrounded by a cupule which is persistent, accrescent, and indurate in fruit (acorn cap). There has been debate over the years regarding the true ontogenetic nature of the cupule. Originally thought to be an involucre of bracts, recent research suggests that the cupule is a complex partial inflorescence derived from stem tissue, see Abbe, 1974;Brett, 1964;Foreman, 1966;MacDonald, 1979;Fey & Endress, 1983. Each cupule subtended by 3 minute, caducous bracteoles.
FEMALE FLOWERS Perianth of one whorl, minute, fragrance absent. Calyx urceolate, of fused sepals. Carpels 3. Locules 3, each containing 2 ovules. Styles 3, each with 1 stigma. Ovary inferior. Placentation axile.
MALE INFLORESCENCES Coetaneous, compound, solitary or fascicled spikes; pendant, catkin-like; in leaf axils of previous year. Rachis densely covered with brown hairs; elongating with age; with 1 subsessile or sessile flower per node, each flower subtended by a small, sessile caducous bracteole.
MALE FLOWERS Perianth of one whorl, fragrance absent. Calyx actinomorphic, campanulate, of fused sepals. Sepal lobes 2-4, broadly oval to ovate with brown hairs moderately dense distributed throughout. Stamens 2-5, exserted. Anthers glabrous, basifixed, opening along the long axis. Filaments free, 1mm long, straight, glabrous. For a review of pollen morphology see Solomon, 1983a.
FRUITS Acorn (glans (Spjut, 1994)) (calybium (Kaul, 1985)) sessile to short-pedunculate; maturation biennial. Acorn ovoid-subglobose, 1-1.7 cm long, comprised of 2 parts- a. the acetabuliform-crateriform-semi-turbinate cup (cupule), enclosing 1/3-1/2 (rarely 3/4) of the base of the nut; and b. the nut, 1-seeded by abortion. For a hypothesis that the first ovule fertilized suppresses the normal development of the others see Mogensen, 1975. Cupule exterior composed of imbricate, appressed scales moderately covered with brown tomentum. Nut olive-green to brown, ovoid-subglobose, with large light-colored circular cupule scar at base and apiculate at the distal end, initially with minute short and unbranched appressed brown to light brown hairs, sparsely distributed throughout, glabrescent, minutely laterally striate.
One study found a size range of .30-1.33 cubic cm with a mean of 0.78 cubic cm and a correlation between smaller acorn size with increasing latitude see Aizen & Woodcock, 1992.
SEEDS Embryo with two large fleshy cotyledons, endosperm lacking. (Young & Young, 1992).
Habitat
Usually occurring in mesic to dry, well-drained substrates of barrens, woods, and roadsides.Distribution
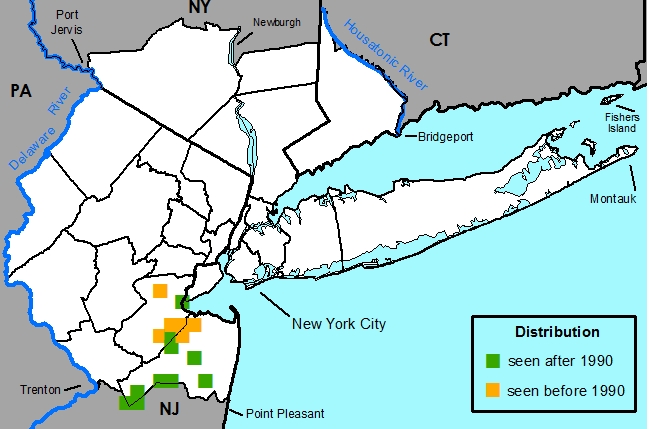
Indigenous to the southeastern United States, extending up the coast to New Jersey.
United States -- AL, AR, DC, DE, FL, GA, IL, IN, KY, LA, MD, MO, MS, NC, NJ, OH, OK, PA, SC, TN, TX, VA, WV
New York Metropolitan Region -- Currently known from only Middlesex and Monmouth Counties, New Jersey.
Rarity Status
Global Heritage Rank -- G5
Connecticut -- Not listed
New Jersey -- Not listed
New York -- Not listed
Species Biology
Flowering
May
Pollination
Anemophily
Fruiting
August [week 1] - October [week 3]
For a study suggesting that masting is effected by weather in conjunction with inherent reproductive cycles see Sork, et al., 1993.
Dispersal
Small predators of acorns facilitate dispersal by dropping undamaged nuts and failing to recover cached nuts. These include Sciurus carolinensis (gray squirrel), Sciurus niger (fox squirrel), Glaucomys volans (Southern Flying Squirrel), Tamias striatus (eastern chipmunk), Peromyscus leucopus (white-footed mouse), Peromyscus maniculatus (deer mouse) and Quiscalus quiscula (common grackle) and Cyanocita cristata (blue jay). (Ivan & Swihart, 2000) (Smith, 1972) (Briggs & Smith, 1989) (Wolff, 1996) (Bosema, 1979) (Darley-Hill & Johnson, 1981) (Johnson & Webb, 1989) (Johnson, et al., 1993)
In addition, one study found that many predators preferred the basal end of the acorn and consumed only 30-60% of the cotyledon. A chemical analyses of acorns from two species revealed that the concentration of protein-precipitable phenolics (primarily tannins) was 12.5% (Q. phellos) and 84.2% (Q. laevis) higher in the apical portion of the seeds where the embryo is located, suggesting that many acorn consumers consistently eat only a portion of the cotyledon of several species of acorns and thereby permit embryo survival. (Steele, et al., 1993).
Probably included in the diet of the grey fox (Urocyon cinereoargenteus) (Scott, 1955), eastern wild turkeys (Meleagris gallopavo silvestris) (Norman, 2003), and white-tailed deer (Odocoileus virginianus) (Bryant, et al., 1996).
Germination
Acorns of members of the black/red oak group have embryo dormancy and germinate naturally the following spring, germination is hypogeal. Moist stratification for 30-90 days at 1-5 d C can be used to break dormancy. For germination to occur the moisture content of the acorns must not drop below 20-30%. It is usually impossible to store acorns for more than 6 months.General rules for collecting and storing acorns: 1. Collect acorns before they lose much water. 2. Ensure acorns are fully hydrated, soak in clean tap water overnight before placing them in storage. 3. Surface dry the acorns just before depositing them in storage to reduce mold growth. 4. Place acorns into cold storage as soon after collection as is possible. (Connor, 2004)
In one study (Q. phellos and Q. laevis), germination experiments revealed equal or greater germination frequencies for partially consumed acorns than for intact acorns. (Steele, et al., 1993).
For a propagation protocol for growing bareroot oaks see Hoss, 2004.